
The British Thermal Unit, commonly known as BTU, is a traditional unit of heat energy. It plays a vital role in heating, ventilation, air conditioning (HVAC) systems, refrigeration, and much more. The BTU measures the heat necessary to increase the temperature of one pound of water by one degree Fahrenheit. This unit of measure is predominantly used in the United States and is a crucial factor in determining the efficiency and capacity of heating and cooling systems.
Delving Deeper into BTU
British Thermal Units, or BTUs, are more than just a unit of measurement. They serve as an indicator of how much heat energy a particular device can produce or a system can handle. The “British” in BTU originates from the traditional British system of units, where the unit was first used. However, BTUs are not only applicable in Britain; they are used worldwide, especially in the fields of heating and cooling.
BTU: Heating Water by 1°F
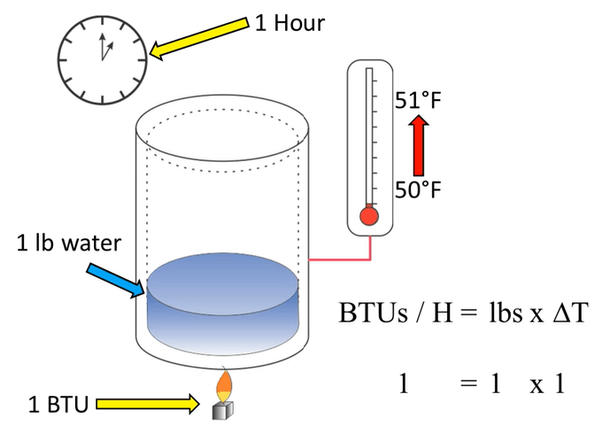
The definition of BTU is rooted in the process of heating water. Specifically, one BTU represents the amount of energy required to raise the temperature of one pound of water by one degree Fahrenheit at sea level. This measurement was established based on the substantial amount of energy required to heat water, making it an ideal standard for measuring thermal energy.
Comparing BTU to Other Units of Heat Measurement
Though BTU is a commonly used unit of heat measurement, it isn’t the only one. The International System of Units (SI) offers an alternative: the kilocalorie. A kilocalorie, often simply called a “calorie” in everyday language, represents the energy required to heat one kilogram of water by one degree Celsius.
The relationship between these two units is quite straightforward: 1 BTU is approximately equal to 0.252 kilocalories. This conversion factor allows us to translate between the traditional system used in the United States and the metric system used in most other countries, enhancing our understanding of heat energy on a global scale.
Concepts Related to BTU
Latent Heat
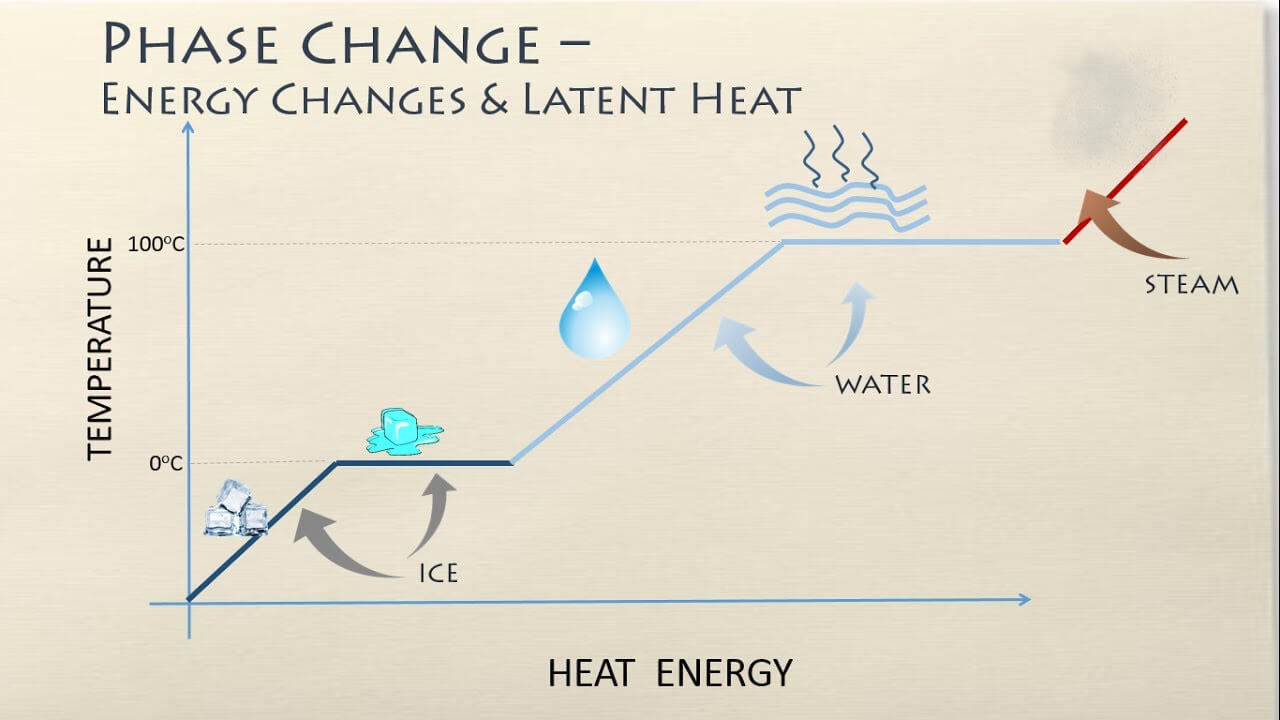
Latent Heat is a concept closely tied to the measurement of BTUs. Essentially, Latent Heat refers to the heat absorbed or released during a phase change of a substance, like the transition of ice to water, without any change in temperature. This absorbed or released energy is measured in BTUs in the United States. For instance, the amount of energy required to transform a pound of ice into water, without raising the temperature, is 144 BTUs. This illustrates the intrinsic connection between the concept of Latent Heat and BTUs as a unit of heat energy measurement.
Sensible Heat
On the other hand, Sensible Heat refers to the heat energy that results in a change of temperature of a substance but not a change in phase. It’s the heat we can “sense” that leads to a noticeable increase or decrease in temperature. For example, if we heat water from 50°F to 60°F, the increase in temperature represents the Sensible Heat, which can also be measured in BTUs. Sensible Heat thus plays a significant role in determining the energy usage of heating and cooling systems.
Rate of Heat Transfer Expressed in BTUs or kW
The rate of Heat Transfer is a crucial concept in understanding thermal dynamics. It describes the amount of heat that moves from one substance to another over a given period, usually expressed in BTUs per hour (Btuh) or kilowatts (kW) for SI units.
For instance, in the field of refrigeration, one common unit of measurement is the ‘ton of refrigeration.’ This term originates from the amount of heat required to melt a ton of ice in a day, which is equivalent to 288,000 BTUs. When divided by the number of hours in a day, this equates to a rate of heat transfer of 12,000 Btuh, underlining the relevance of BTUs in everyday applications of heat transfer.
Conversion Calculator Between BTU and USRT(A Ton of Refrigeration)
Learn more about USRT at What is USRT?
The Use of BTU in Everyday Life
The concept of BTUs may seem abstract and purely scientific, but it’s integral to many aspects of our everyday lives, particularly in heating, cooling, and refrigeration systems.
BTU in Heating and Cooling Systems

In the context of heating and cooling systems, BTUs provide a standardized measurement for the amount of heat energy a system can remove (for cooling) or add (for heating) from a space per hour. This is critical when it comes to choosing the correct size of a heating or cooling system for a particular space. For example, an air conditioner’s cooling capacity is often specified in BTUs. A higher BTU rating means a greater cooling capacity, which can cool a larger room more effectively.
BTU in Refrigeration

Similarly, in refrigeration, BTUs play an essential role in quantifying the performance of refrigeration systems. This is where the concept of ‘a ton of refrigeration’ comes in. As explained earlier, a ‘ton of refrigeration’ is defined as the amount of heat required to melt a ton (2,000 lbs) of ice in a 24-hour period. This equates to a rate of heat transfer of 12,000 BTUs per hour. Therefore, when you see a refrigeration system rated in tons, it’s a direct reference to its capacity to remove heat, measured in BTUs. This standard of measurement allows for consistency and ease of comparison when selecting and evaluating refrigeration systems.
Heat Transfer Formula and BTU
An essential formula used across various scientific and engineering disciplines is the heat transfer formula. It is represented as Q = M x Cp x ΔT. In this formula:
- Q stands for the heating or cooling capacity and is expressed in BTUs per hour.
- M is the mass of the fluid per hour, typically in pounds per hour.
- Cp represents the specific heat of the fluid (for water, it’s 1 BTU/lb/°F).
- ΔT is the temperature difference between the entering and leaving fluid in degrees Fahrenheit.
This equation showcases the interrelation between BTU and heat transfer. BTU, as a measure of energy, gives a quantifiable value to Q, the heating or cooling capacity. The heat transfer formula further simplifies for water to become Btu/hr = GPM x 500 x ΔT, emphasizing BTU’s role in practical heating and cooling calculations.
Conclusion
BTUs represent a fundamental unit in understanding and measuring heat energy. From heating and cooling our homes to industrial processes, BTUs play a pivotal role in our daily lives and the broader scientific community. A deeper exploration of BTUs and their associated concepts can aid in everything from personal decisions about home appliances to larger-scale energy efficiency efforts. By understanding the value and wide application of BTUs, we are better equipped to navigate the world of heat transfer and energy conservation.
FAQs
Q1: What exactly is a British Thermal Unit (BTU)?
A BTU is a traditional unit of heat energy used primarily in the United States. It represents the amount of heat necessary to increase the temperature of one pound of water by one degree Fahrenheit. This measure is critical in determining the capacity and efficiency of heating and cooling systems.
Q2: How does the BTU compare to other units of heat measurement like the kilocalorie?
The BTU and the kilocalorie are both measures of heat, but they stem from different systems. While the BTU is widely used in the United States, the kilocalorie is an SI (International System) unit. The conversion factor between the two is fairly straightforward: 1 BTU is equivalent to approximately 0.252 kilocalories.
Q3: What is the role of BTU in the heating and cooling systems?
In heating and cooling systems, BTUs serve as a standardized measure for the amount of heat energy a system can remove (for cooling) or add (for heating) to a space per hour. This makes BTUs instrumental in choosing the correct size of heating or cooling systems, ensuring that they can adequately meet a room’s specific heating and cooling requirements.
Q4: How does the BTU feature in the heat transfer formula?
The heat transfer formula is Q = M x Cp x ΔT. In this formula, ‘Q’ stands for the heating or cooling capacity, which is expressed in BTUs per hour. This showcases the interrelation between BTU, a measure of energy, and the heat transfer process.
Q5: Why are BTUs important in the field of energy conservation?
Understanding BTUs can significantly aid in energy conservation and efficiency efforts. BTU ratings of appliances provide insights into their energy usage, thereby enabling more informed decisions regarding their operation. This can lead to more energy-efficient homes and lower energy costs.














