Solvent recovery systems are designed to capture, condense, and reuse valuable solvents that would otherwise be lost to the atmosphere or sent to waste treatment. In chemical processing, pharmaceuticals, coatings, printing, adhesives, and specialty manufacturing, this is both an economic and environmental requirement. But solvent recovery only works efficiently when the vapor stream is cooled quickly and safely—and that’s where industrial chillers become a core part of the system.
Unlike ordinary process cooling, solvent recovery cooling must deal with volatile, flammable, and often corrosive media. The chiller isn’t just removing heat—it’s helping control vapor pressure, improve condensation efficiency, stabilize recovery yield, and reduce fire and explosion risk. In practice, the cooling system becomes part of the safety architecture of the entire plant.
For that reason, the best industrial chiller for solvent recovery systems is usually not a generic water chiller. It’s a purpose-built unit with the right refrigeration capacity, hazardous-area protection, corrosion-resistant materials, and a thermal design matched to the specific solvent being recovered.
Why Cooling Is Critical in Solvent Recovery

A solvent recovery system typically works by heating a solvent mixture, separating the volatile solvent from impurities, and then condensing the vapor back into liquid form. This process is thermodynamically simple in principle, but in practice it’s highly sensitive to temperature.
If the condenser is too warm, vapor won’t liquefy efficiently. Recovery rate drops, emissions increase, and the system has to run longer to achieve the same output. If the condenser is too cold or the cooling response is unstable, the system may experience pressure fluctuation, uneven reflux, or unnecessary energy use. In systems that handle low-boiling solvents such as acetone (bp 56°C), ethanol (bp 78°C), MEK (bp 80°C), or toluene (bp 111°C), the margin for error is small because vapor can accumulate quickly and create an ignition hazard.
That’s why cooling performance directly affects three things at once: recovery efficiency, product purity, and operational safety.
Important safety point: In solvent recovery, cooling is not only a process function—it is also a fire-risk control measure.
How a Solvent Recovery System Works
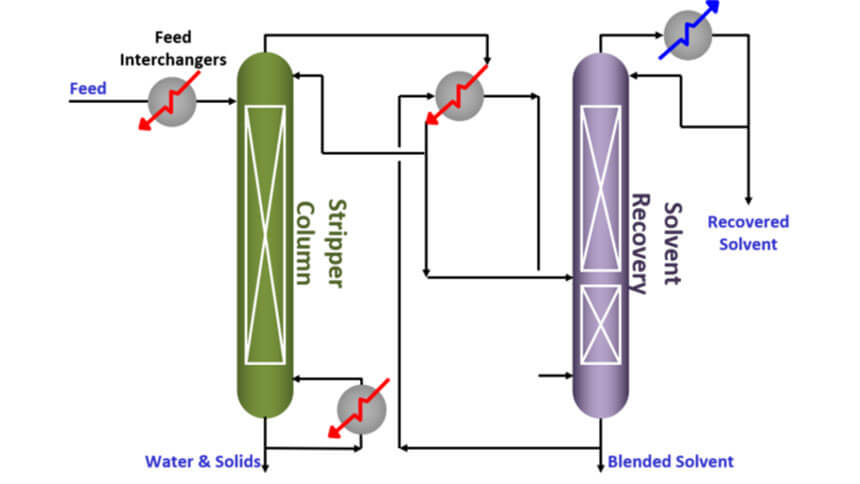
Most recovery systems follow the same basic sequence. The solvent-bearing feed is heated in a still, evaporator, distillation vessel, or vacuum chamber. As the liquid boils, solvent vapor rises and passes into a condenser or cooling section. There, the industrial chiller supplies chilled fluid to remove latent heat from the vapor stream. The vapor condenses into liquid, which is collected for reuse. Non-condensable gases are vented through a controlled system.
Q = ṁ × (hvapor − hliquid) = ṁ × hfgWhere ṁ = vapor mass flow rate, hfg = latent heat of vaporizationContoh: Acetone hfg ≈ 518 kJ/kg, Ethanol hfg ≈ 846 kJ/kg
Same mass flow → ethanol needs ~63% more cooling than acetone
What determines overall performance isn’t simply the boiling point of the solvent—it’s the relationship between vapor load, condenser surface area, coolant temperature, and system pressure. If any one of those variables is mismatched, recovery efficiency falls.
In more advanced systems, the chiller is tied to a reflux loop, vacuum system, or multi-stage heat exchanger network. This allows the plant to lower boiling temperature, improve solvent separation, and reduce thermal stress on both the solvent and the equipment.
Why Industrial Chillers Are the Right Cooling Source
Industrial chillers are preferred because they provide stable, controllable, continuous cooling under industrial load. A properly engineered chiller can hold coolant temperature within a narrow band even when the vapor load changes. That stability matters because solvent recovery loads are often cyclical—a batch distillation run may start slowly, peak during active boiling, then taper off. The chiller must follow that profile without overshooting or short cycling.
A good chiller also protects downstream equipment. Condensers, piping, seals, and valves last longer when the coolant temperature is steady. In contrast, unstable cooling leads to pressure spikes, uneven condensation, and repeated mechanical stress—basically, everything wears out faster.
In solvent recovery plants, the chiller is often part of a closed-loop system with one loop on the refrigeration side and another on the process side. That separation keeps the cooling medium isolated from the solvent stream and allows the system to use corrosion-resistant materials and safer operating fluids.
The Main Components of an Industrial Solvent Recovery Chiller
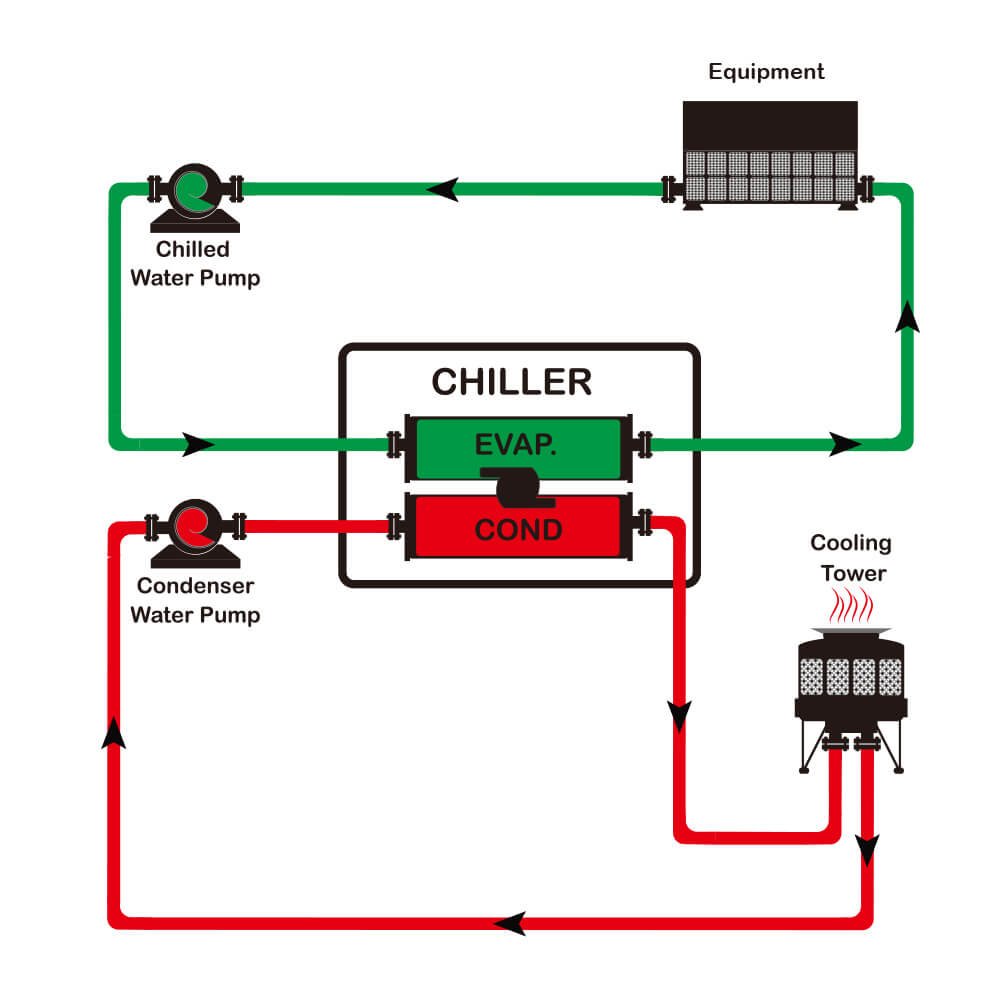
A solvent recovery chiller is built from several subsystems, and each one matters.
The compressor is the energy engine of the machine. In smaller systems, scroll compressors are often used because they’re compact and reliable. In larger systems, screw compressors are common because they handle heavier loads and offer better modulation. For large centralized plants, centrifugal compressors may be used where capacity is very high and steady-state efficiency matters. The right choice depends on recovery load, duty cycle, and plant scale.
The condenser is where heat leaves the refrigerant. Water-cooled condensers generally deliver better efficiency on larger systems because water transfers heat more effectively than air. Air-cooled condensers, on the other hand, simplify installation and are often better for smaller plants or locations without cooling tower infrastructure.
The evaporator is where the process fluid is cooled—typically a plate heat exchanger or shell-and-tube exchanger. A well-designed evaporator improves heat transfer rate and reduces temperature swing at the condenser or recovery vessel.
The expansion valve meters refrigerant flow into the evaporator. Electronic expansion valves are especially useful in solvent recovery because they respond quickly to changing loads and maintain stable superheat—important when vapor flow changes rapidly during distillation.
The pump system moves coolant through the process loop. In larger systems, variable-frequency pumps are preferred because they adjust to changing demand and reduce energy consumption.
The control system coordinates the whole unit. Modern industrial chillers use PLC-based control, temperature sensors, pressure transmitters, flow switches, and fault logic to manage capacity and protect the system. In solvent recovery service, automation is especially important because it reduces operator exposure to hazardous materials and keeps the process stable.
Explosion-Proof Design Is Not Optional

Solvent recovery often deals with flammable or volatile organic compounds. That means the cooling system must be designed for hazardous environments, not standard utility space.
A proper solvent recovery chiller may need explosion-proof electrical components, certified control enclosures, and hazard-rated wiring and motors. Depending on the site classification, the system may need to comply with ATEX (Europe), IECEx (international), or Class I Division I (North America) requirements. The exact standard depends on region, solvent class, and the area classification of the installation.
The practical engineering goal is simple: prevent the cooling system from becoming an ignition source. That means controlling electrical arc risk, hot surface risk, static accumulation, and leakage pathways.
This is especially important in solvent recovery installations because vapor can appear at low points, around seals, and near venting areas. The chiller should therefore be located and configured so that it doesn’t expose electrical parts or hot components to solvent vapor.
Choosing the Right Refrigerant

Refrigerant selection matters because it affects efficiency, safety, and regulatory compliance.
| Pendingin | GWP | Kelas Keamanan | Best For |
|---|---|---|---|
| R134a | 1430 | A1 (non-flammable) | Legacy systems, replacement |
| R513A | 573 | A1 (non-flammable) | R134a retrofit, new systems |
| R1234ze | 1 | A2L (mildly flammable) | New low-GWP designs |
| R290 (propane) | 3 | A3 (flammable) | Maximum environmental performance |
| CO₂ (R744) | 1 | A1 (non-flammable) | High-pressure applications |
For systems where maximum environmental performance is required, natural refrigerants such as R290 or CO₂ may be considered. However, they come with design constraints—R290 is highly flammable and requires tight charge limits, while CO₂ operates at much higher pressures, which increases equipment complexity.
The best refrigerant depends on the solvent load, ambient conditions, installation standard, and the plant’s risk tolerance. For hazardous solvent recovery, safety class and refrigerant charge management are often just as important as COP.
Coolant Medium and Process Loop Design
The process-side coolant is usually water, glycol-water mixture, or another heat transfer medium selected for temperature range and freeze protection.
If the system only needs moderate cooling above freezing, chilled water is often the simplest and most efficient option. If the process must operate below 0°C or in colder ambient conditions, glycol is typically added to prevent freezing. The glycol concentration needs to be balanced carefully—too much glycol reduces heat capacity and increases pumping losses, so it’s a trade-off worth getting right.
In solvent recovery, the coolant loop should be closed, filtered, and corrosion-controlled. Most industrial systems favor stainless steel, copper-free piping where necessary, and seals compatible with the coolant chemistry. For aggressive solvent environments, 316L stainless steel, PTFE-compatible seals, and chemically resistant gaskets are common choices.
The loop must also be designed for stable turbulence. If flow is too low, heat transfer efficiency drops. If it’s too high, pressure loss rises and pump energy increases. The goal is a hydraulic design that maintains steady, repeatable heat removal at the condenser.
Air-Cooled vs Water-Cooled Chillers for Solvent Recovery

| Item | Berpendingin Udara | Berpendingin Air |
|---|---|---|
| Instalasi | Simpler, no water infrastructure | Requires cooling tower or dry cooler |
| Energy Efficiency (COP) | 3.0–4.5 | 4.0–6.0 |
| Stabilitas suhu | Good (±0.3–0.5°C) | Excellent (±0.1–0.3°C) |
| Ambient Sensitivity | High (5–8% per 10°C rise) | Low (2–3% per 10°C rise) |
| Best For | Small to medium plants | Large continuous-duty plants |
Air-cooled systems are easier to install because they don’t require cooling towers or condenser water treatment. They’re often the better choice for smaller solvent recovery lines, pilot plants, and facilities where quick installation matters.
Water-cooled systems are usually better for larger or continuous-duty applications. Water has much higher heat transfer capacity than air, so water-cooled systems can maintain lower condensing temperatures and higher efficiency. In high-load operation, that usually means better energy performance and more stable recovery.
As a rule of thumb: air-cooled for smaller and moderate loads, water-cooled as the duty rises and the plant runs longer hours.
How to Size a Solvent Recovery Chiller

Sizing the chiller correctly is one of the most important steps in the design process. The cooling load depends on latent heat of vaporization, feed temperature, solvent mixture composition, reflux rate, condenser efficiency, and any heat recovery arrangement.
A practical engineering approach is to estimate the vapor load first, then add sensible heat from the feed and a safety margin for process variation. If the solvent has a high vaporization load, the condenser duty can rise very quickly.
For example, a system recovering acetone or ethanol may need significantly different cooling behavior even when the liquid volume seems similar, because their vapor pressure and heat of vaporization differ. A low-boiling solvent will often need faster and stronger heat rejection than a higher-boiling one.
The design margin should not be tight. Solvent recovery systems benefit from extra cooling headroom because load can rise unexpectedly during startup, vacuum changes, or batch transition. Plan for the worst, and the system will handle everything else gracefully.
Selection Criteria That Matter Most
When specifying an industrial chiller for solvent recovery, the most important criteria are:
- Cooling capacity matched to peak vapor load
- Coolant supply temperature stability
- Refrigerant safety class and charge control
- Explosion-proof electrical design
- Corrosion-resistant wetted materials
- Pump stability and flow control
- Compressor modulation range
- Maintenance access and uptime strategy
Those factors matter more than catalog horsepower alone. A chiller that looks strong on paper can still fail in solvent service if its controls are unstable, its materials are wrong, or its safety classification is insufficient.
Energy Recovery and Process Integration

Solvent recovery systems often release a lot of heat during condensation. In some plants, that heat can be partially recovered for other uses, such as preheating feed, supporting another process loop, or supplying hot water for auxiliary operations.
A heat recovery chiller can be a useful option where the plant has both cooling and heating demand. Instead of rejecting all condenser heat to the ambient environment, part of that heat is redirected into a hot water loop. This can improve total plant energy performance.
That said, heat recovery only makes sense when the plant can actually use the recovered heat. If there’s no useful heat sink, a standard chiller arrangement may be simpler and more cost-effective. Don’t overcomplicate things for the sake of it.
Maintenance and Reliability
Solvent recovery chillers need routine inspection because the operating environment is demanding. The condenser should be kept clean, the coolant loop should be filtered, and the compressor oil condition should be monitored. Sensor drift, refrigerant leakage, and fouling in the heat exchanger can all reduce performance over time.
A good maintenance routine should include inspection of:
- Coolant level and concentration
- Compressor suction and discharge pressure
- Condenser and evaporator cleanliness
- Electrical connections and enclosure seals
- Valve response and control logic
- Pump vibration and flow performance
- Leak detection and alarm functions
In hazardous solvent environments, preventive maintenance isn’t just about uptime—it’s also a safety requirement.
Kesimpulan
Industrial chillers are a core technology in solvent recovery systems because they control condensation, stabilize pressure, improve recovery yield, and reduce fire risk. The right chiller must do more than cool—it must work safely in hazardous conditions, maintain stable performance under changing vapor loads, and integrate cleanly with the recovery process.
For smaller installations, air-cooled systems offer simplicity and flexibility. For larger plants, water-cooled systems deliver better efficiency and stability. For any facility handling flammable or volatile solvents, explosion-proof design, correct refrigerant selection, and proper material compatibility are non-negotiable.
In solvent recovery, cooling is not a background utility. It is a process and safety system at the same time—and getting it right makes everything else work better.
